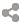Radon
(#35507178)
Level 1 Imperial
Click or tap to view this dragon in Predict Morphology.
Energy: 0/50

Expand the dragon details section.
Collapse the dragon details section.
Personal Style
Apparel



Skin
Scene
Measurements
Length
25.87 m
Wingspan
18.4 m
Weight
7046.23 kg
Genetics
Radioactive
Jaguar
Jaguar
Radioactive
Rosette
Rosette
Leaf
Underbelly
Underbelly
Hatchday
Breed
Eye Type
Level 1 Imperial
EXP: 0 / 245


STR
6
AGI
6
DEF
6
QCK
5
INT
8
VIT
8
MND
6
Biography
RADON BASIC FACTS
Atomic Number: 86
Symbol: Rn
Atomic Weight: 222.0176
Discovery: Fredrich Ernst Dorn 1898 or 1900 (Germany), discovered the element and called it radium emanation. Ramsay and Gray isolated the element in 1908 and named it niton.
Electron Configuration: [Xe] 4f14 5d10 6s2 6p6
Word Origin: from radium. Radon was once called niton, from the Latin word nitens, which means 'shining'
Isotopes: At least 34 isotopes of radon are known ranging from Rn-195 to Rn-228.
There are no stable isotopes of radon. The isotope radon-222 is the most stable isotope and called thoron and emanates naturally from thorium. Thoron is an alpha-emitter with a half-life of 3.8232 days. Radon-219 is called actinon and emanates from actinium. It is an alpha-emitter with a half-life of 3.96 sec.
Properties: Radon has a melting point of -71°C, boiling point of -61.8 °C, gas density of 9.73 g/l, specific gravity of the liquid state of 4.4 at -62°C, specific gravity of the solid state of 4, usually with a valence of 0 (it does form some compounds, however, such as radon fluoride). Radon is a colorless gas at normal temperatures. It is also the heaviest of the gases. When it is cooled below its freezing point it displays a brilliant phosphorescence. The phosphorescence is yellow as the temperature is lowered, becoming orange-red at the temperature of liquid air. Inhalation of radon presents a health risk.
Radon build-up is a health consideration when working with radium, thorium, or actinium. It is also a potential issue in uranium mines.
Sources: It is estimated that each square mile of soil to a depth of 6 inches contains approximately 1 g of radium, which releases radon to the atmosphere. The average concentration of radon is about 1 sextillion parts of air.
Radon naturally occurs in some spring waters.
Element Classification: Inert Gas
Atomic Number: 86
Symbol: Rn
Atomic Weight: 222.0176
Discovery: Fredrich Ernst Dorn 1898 or 1900 (Germany), discovered the element and called it radium emanation. Ramsay and Gray isolated the element in 1908 and named it niton.
Electron Configuration: [Xe] 4f14 5d10 6s2 6p6
Word Origin: from radium. Radon was once called niton, from the Latin word nitens, which means 'shining'
Isotopes: At least 34 isotopes of radon are known ranging from Rn-195 to Rn-228.
There are no stable isotopes of radon. The isotope radon-222 is the most stable isotope and called thoron and emanates naturally from thorium. Thoron is an alpha-emitter with a half-life of 3.8232 days. Radon-219 is called actinon and emanates from actinium. It is an alpha-emitter with a half-life of 3.96 sec.
Properties: Radon has a melting point of -71°C, boiling point of -61.8 °C, gas density of 9.73 g/l, specific gravity of the liquid state of 4.4 at -62°C, specific gravity of the solid state of 4, usually with a valence of 0 (it does form some compounds, however, such as radon fluoride). Radon is a colorless gas at normal temperatures. It is also the heaviest of the gases. When it is cooled below its freezing point it displays a brilliant phosphorescence. The phosphorescence is yellow as the temperature is lowered, becoming orange-red at the temperature of liquid air. Inhalation of radon presents a health risk.
Radon build-up is a health consideration when working with radium, thorium, or actinium. It is also a potential issue in uranium mines.
Sources: It is estimated that each square mile of soil to a depth of 6 inches contains approximately 1 g of radium, which releases radon to the atmosphere. The average concentration of radon is about 1 sextillion parts of air.
Radon naturally occurs in some spring waters.
Element Classification: Inert Gas
Click or tap a food type to individually feed this dragon only. The other dragons in your lair will not have their energy replenished.
Feed this dragon Insects.
Feed this dragon Meat.
Feed this dragon Seafood.
Feed this dragon Plants.
Exalting Radon to the service of the Icewarden will remove them from your lair forever. They will leave behind a small sum of riches that they have accumulated. This action is irreversible.
Do you wish to continue?
- Names must be longer than 2 characters.
- Names must be no longer than 16 characters.
- Names can only contain letters.
- Names must be no longer than 16 characters.
- Names can only contain letters.