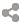Francium
(#35471825)
Level 1 Spiral
Click or tap to view this dragon in Predict Morphology.
Energy: 0/50

Expand the dragon details section.
Collapse the dragon details section.
Personal Style
Apparel



Skin
Scene
Measurements
Length
3.94 m
Wingspan
2.52 m
Weight
73.72 kg
Genetics
Radioactive
Jaguar
Jaguar
Radioactive
Rosette
Rosette
Radioactive
Underbelly
Underbelly
Hatchday
Breed
Eye Type
Level 1 Spiral
EXP: 0 / 245


STR
5
AGI
9
DEF
5
QCK
8
INT
6
VIT
6
MND
6
Biography
FRANCIUM BASIC FACTS
Atomic Number: 87
Symbol: Fr
Atomic Weight: 223.0197
Discovery: Discovered in 1939 by Marguerite Perey of the Curie Institute, Paris (France).
Electron Configuration: [Rn] 7s1
Word Origin: Named for France, the country of its discoverer.
Isotopes: There are 33 known isotopes of francium. The longest lived is Fr-223, a daughter of Ac-227, with a half-life of 22 minutes. This is the only naturally-occuring isotope of francium.
Properties: The melting point of francium is 27 °C, boiling point is 677 °C, and its valence is 1. Francium is the heaviest known member of the alkali metals series. It has the highest equivalent weight of any element and is the most unstable of the first 101 elements of the periodic system. All known isotopes of francium are highly unstable, so knowledge of the chemical properties of this element comes from radiochemical techniques. No weighable quantity of the element has been prepared or isolated. The chemical properties of francium most closely resemble those of cesium.
Sources: Francium occurs as a result of an alpha disintegration of actinium. It can be produced by artificially bombarding thorium with protons. It occurs naturally in uranium minerals, but there is probably less than an ounce of francium at any time in the total crust of the earth.
Element Classification: alkali Metal
Atomic Number: 87
Symbol: Fr
Atomic Weight: 223.0197
Discovery: Discovered in 1939 by Marguerite Perey of the Curie Institute, Paris (France).
Electron Configuration: [Rn] 7s1
Word Origin: Named for France, the country of its discoverer.
Isotopes: There are 33 known isotopes of francium. The longest lived is Fr-223, a daughter of Ac-227, with a half-life of 22 minutes. This is the only naturally-occuring isotope of francium.
Properties: The melting point of francium is 27 °C, boiling point is 677 °C, and its valence is 1. Francium is the heaviest known member of the alkali metals series. It has the highest equivalent weight of any element and is the most unstable of the first 101 elements of the periodic system. All known isotopes of francium are highly unstable, so knowledge of the chemical properties of this element comes from radiochemical techniques. No weighable quantity of the element has been prepared or isolated. The chemical properties of francium most closely resemble those of cesium.
Sources: Francium occurs as a result of an alpha disintegration of actinium. It can be produced by artificially bombarding thorium with protons. It occurs naturally in uranium minerals, but there is probably less than an ounce of francium at any time in the total crust of the earth.
Element Classification: alkali Metal
Click or tap a food type to individually feed this dragon only. The other dragons in your lair will not have their energy replenished.
Feed this dragon Insects.
Feed this dragon Meat.
This dragon doesn't eat Seafood.
This dragon doesn't eat Plants.
Exalting Francium to the service of the Icewarden will remove them from your lair forever. They will leave behind a small sum of riches that they have accumulated. This action is irreversible.
Do you wish to continue?
- Names must be longer than 2 characters.
- Names must be no longer than 16 characters.
- Names can only contain letters.
- Names must be no longer than 16 characters.
- Names can only contain letters.