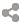Enthalpy
(#76963378)
Level 1 Gaoler
Click or tap to view this dragon in Predict Morphology.
Energy: 50/50

Expand the dragon details section.
Collapse the dragon details section.
Personal Style

Ancient dragons cannot wear apparel.
Skin
Scene
Measurements
Length
12.76 m
Wingspan
7.84 m
Weight
9542.49 kg
Genetics
Ice
Wasp (Gaoler)
Wasp (Gaoler)
Ice
Bee (Gaoler)
Bee (Gaoler)
Ice
Opal (Gaoler)
Opal (Gaoler)
Hatchday
Breed
Eye Type
Level 1 Gaoler
EXP: 0 / 245


STR
7
AGI
5
DEF
7
QCK
5
INT
5
VIT
9
MND
7
Biography
Enthalpy
Enthalpy /ˈɛnθəlpi/ (listen), a property of a thermodynamic system, is the sum of the system's internal energy and the product of its pressure and volume. It is a state function used in many measurements in chemical, biological, and physical systems at a constant pressure, which is conveniently provided by the large ambient atmosphere. The pressure–volume term expresses the work required to establish the system's physical dimensions, i.e. to make room for it by displacing its surroundings. The pressure-volume term is very small for solids and liquids at common conditions, and fairly small for gases. Therefore, enthalpy is a stand-in for energy in chemical systems; bond, lattice, solvation and other "energies" in chemistry are actually enthalpy differences. As a state function, enthalpy depends only on the final configuration of internal energy, pressure, and volume, not on the path taken to achieve it.
Enthalpy /ˈɛnθəlpi/ (listen), a property of a thermodynamic system, is the sum of the system's internal energy and the product of its pressure and volume. It is a state function used in many measurements in chemical, biological, and physical systems at a constant pressure, which is conveniently provided by the large ambient atmosphere. The pressure–volume term expresses the work required to establish the system's physical dimensions, i.e. to make room for it by displacing its surroundings. The pressure-volume term is very small for solids and liquids at common conditions, and fairly small for gases. Therefore, enthalpy is a stand-in for energy in chemical systems; bond, lattice, solvation and other "energies" in chemistry are actually enthalpy differences. As a state function, enthalpy depends only on the final configuration of internal energy, pressure, and volume, not on the path taken to achieve it.
Click or tap a food type to individually feed this dragon only. The other dragons in your lair will not have their energy replenished.
This dragon doesn't eat Insects.
Feed this dragon Meat.
This dragon doesn't eat Seafood.
Feed this dragon Plants.
Exalting Enthalpy to the service of the Lightweaver will remove them from your lair forever. They will leave behind a small sum of riches that they have accumulated. This action is irreversible.
Do you wish to continue?
- Names must be longer than 2 characters.
- Names must be no longer than 16 characters.
- Names can only contain letters.
- Names must be no longer than 16 characters.
- Names can only contain letters.